
That’s because chemistry deals with physical matter, and often needs to separate one substance from another.
So I came to chemistry lab and chose my station. And then I looked up and saw this:

That’s the ceiling. Clearly, someone had an interesting (read: explosive. The ceiling is a good twelve to fifteen feet above my head) chemical reaction. A reminder, or maybe a hope, that something like that could happen to me.
The lab I took pictures of is an isolation of caffeine from tea. So the first thing we did was a good, hot cup of tea. This being chemistry, we have to do it with beakers and hot plates, and there’s just a horrible amount of stuff hanging around, including an ice bath and a filter and vile stuff (how vile? Just wait, I’ve got a shot of the label of one of the ingredients we used) that we’re going extract the caffeine with.

Having boiled the tea to get all the fascinating organic compounds out of it, we add a binding agent. Helpful hint to those of you who have not taken chemistry: hot glass looks exactly like cold glass. If you don’t know what temperature the glass is, put your hand near it first.

Having brewed tea, and added chemicals to make it completely undrinkable (the modern method for extracting caffeine from tea involves dipping it in high-pressure liquid carbon dioxide, rather than using a ethyl acetate, which is why decaffeinated tea and coffee no longer taste like ethyl acetate), we cool it in an ice bath.

We then filter the result into a Buchner flask. This removes some of what we added to the tea. The hose is a vacuum pump attached to the water faucet, more to encourage the tea through the filter than anything else.

Now we get to the cool glassware. The shape of the seperatory funnel helps separate the liquid by density. The denser stuff goes to the bottom, and the lighter stuff can sit on the top. You can see it beginning to separate in the photo. We use the stopcock to pour the clear stuff out the bottom, while making sure we don’t get any of the tea-colored liquid. We end up with a small amount of clear liquid in to a beaker. We then set this under the hood and leave it to evaporate.
The rest of the class comes back in a week. I spy on it the next day. The liquid has evaporated, leaving behind a chalky white substance.

We think it’s caffeine, but how can we be sure? And how will we be able to remove any impurities? The answer appears to be a crack pipe.

But it’s not really a crack pipe. It’s a complex piece of glassware called a cold finger. We shove ice down the top of it which has a long extrusion into the bottom cavity. So we can heat something and provide it with a cold surface to reform onto after it has sublimated. Caffeine will burn off the crystals we have and stick to the cold finger, where the impurities shouldn’t. The stem to the side will be attached to a vacuum pump so that this can be achieved at low pressure.

Here you can see the ice going down the cold finger, and there is our collection of suspicious white crystal at the bottom. And it’s resting on a hot plate. I swear that the rubber hose is not feeding into someone’s lungs.

See?

The reason we all wear safety goggles is because we’re noodling around with stuff like this. No wonder Peter Parker got his fame from being in a science lab. Where else are you going to find an off-the-shelf mutagen?

After weighing how much stuff we have scraped off the interior of the

This is a 10 mL beaker, and there might actually be 1 mL in it. If Barbie ever does science in her Malibu Dream College, she’ll come with an assortment of 10 mL beakers. Probably in fabulous colors.

This piece of equipment was designed by a sadist. It’s ostensibly so that you can look at the eensy weensy sample you’ve put in this machine and see exactly when it melts. The melting point of something is a good indication that it is what you think it is. But that’s not the fun part.
You see that yellow sticker? It says “Caution Unit Hot.” Do you know what’s directly above that sticker? The eyepiece. Will the chemistry student be able to look through the eyepiece without burning the shit out of their cheeks? Ho ho ho! Science has a sense of humor!
There is another way to check to make sure that the caffeine we’ve labored over so hard is in fact caffeine. With a Nuclear Magnetic Resonator.

One of these. An advantage to taking chemistry at Bennington College is that they are proud of their science program, and are willing to plunk down the 300 large it takes to own Nuclear Magnetic Resonator. And the additional 12 K per hear it takes to feed the thing helium coolant. So at the end of this lab, yes, I got to drive the school’s NMR.
Now, interpreting the results was surprisingly difficult for something so damn advanced. The NMR tells you about the molecules you’re dealing with indirectly. Your results are based on the hydrogen atoms that are bonded to the atoms next to the one you’re looking at. Is that clear? So it will tell you, for example, that you’ve got eight hydrogens near a particular nucleus, and you have to parse out what atoms they’re near and how many go on each one. For the whole chain. It’s surprisingly like a cryptography sequence.
So at the end, hurrah, we have managed to isolate this stuff:
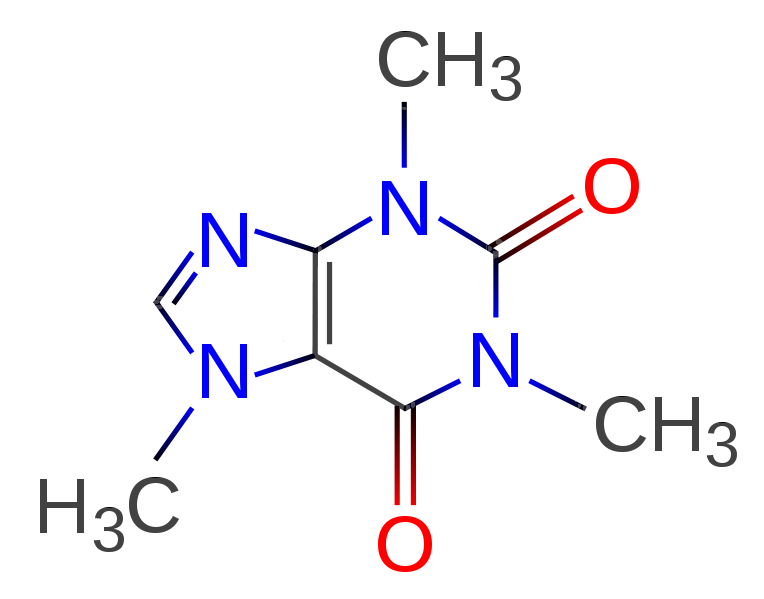
This is a sequence of three consecutive labs, all of which built on each other. I figured if I was going to photograph and share, I might as well share the most impressive-looking one.
Tomorrow, the Good News.
No comments:
Post a Comment